Sterile vs. Non-Sterile Gloves
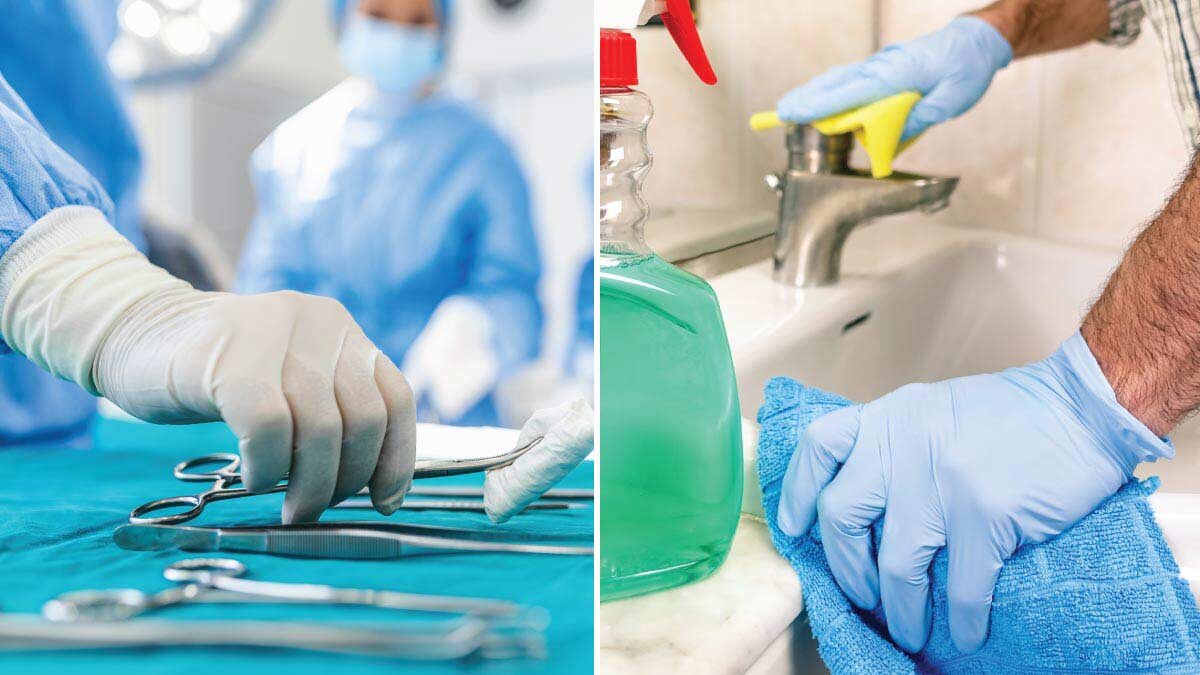
Whether you run a small clinic, oversee a food prep team, or manage a light manufacturing operation, choosing the right type of disposable gloves helps protect your employees and maintain compliance with safety regulations. Two of the most common options are sterile and non-sterile gloves. Understanding how they differ can help your business meet regulatory expectations and support hygiene practices across a range of tasks. Let’s break it down in a way that makes sense for your team.
What Are Sterile Gloves?
Sterile gloves are manufactured under strict conditions to eliminate all forms of microbial life. They’re typically sterilized using methods like gamma radiation or ethylene oxide, and each pair is individually wrapped to maintain sterility until use.
These gloves are often required in:
- Medical and surgical procedures
- Sterile field operations (such as catheter insertion or wound care)
- Cleanroom environments like pharmaceutical labs or research facilities
If your business operates in healthcare or another field that involves contact with sterile environments or body entry points, sterile gloves may be necessary to meet industry-specific protocols.
What Are Non-Sterile Gloves?
Non-sterile gloves, also known as exam gloves or general-purpose gloves, are clean but not sterilized. They’re widely used across industries that don’t require a completely sterile environment.
These gloves are a go-to choice for:
- Routine patient care in non-surgical settings
- Food preparation and service
- Janitorial or custodial work
- Light manufacturing and assembly tasks with low contamination risk
If you're focused on day-to-day operations where sterility isn’t essential, non-sterile gloves offer a practical and cost-effective solution.
OSHA and FDA Guidelines: What You Need to Know
When it comes to glove selection, here’s how federal guidelines apply.
OSHA doesn’t mandate sterile gloves specifically. However, under 29 CFR 1910 Subpart I, employers must provide personal protective equipment (PPE) appropriate for the job. That means conducting a hazard assessment and choosing gloves that match the level of risk.
FDA regulations apply if your business handles medical, surgical, or food-related activities. Sterile gloves are typically required in surgical and high-risk medical scenarios. For food handling, gloves must meet cleanliness standards but don’t necessarily need to be sterile.
When Should You Use Each?
Here’s a simple side-by-side to guide your decision.
| Use Case | Recommended Glove Type |
| Invasive medical or surgical procedures | Sterile |
| Handling open wounds or sterile fields | Sterile |
| Routine exams or patient interactions | Non-Sterile |
| Food prep or general manufacturing | Non-Sterile |
| Cleaning, maintenance, or janitorial work | Non-Sterile |
Choosing between sterile and non-sterile gloves isn’t just about safety; it’s also about matching the glove to the task so you’re using your resources wisely. For small business owners, this choice can affect compliance, budget, and daily workflow. Keeping both types of gloves in stock may offer the flexibility your team needs. And if you're ever unsure, reviewing your PPE policy or conducting a fresh hazard assessment can provide clarity.
Product Compliance and Suitability
The statements contained in this guide are intended for general informational purposes only. Such statements do not constitute a product recommendation or representation as to the appropriateness, accuracy, completeness, correctness, or currentness of the information provided. Information provided in this guide does not replace the use by you of any manufacturer instructions, technical product manual, or other professional resource or adviser available to you. Always read, understand, and follow all manufacturer instructions. Portions of this article were generated in part by ChatGPT, and edited by a member of the Zoro team.
